TPD Ready E-liquids
With new regulations coming into effect in Europe, the e-cigarette industry is now entering a new, more professional era. Many producers will disappear from the market unable to comply with such strict restrictions on e-liquids. Ritchy is one of the largest producers of e-liquids and we respect the highest quality stadards in the e-cigarette industry. Our e-liquids are TPD compliant and notified.
How the new laws affect vaping industry?
According to the new EU TPD regulation that came into force on 1st January 2017, e-liquids must meet the following requirements:
- The strength of nicotine in a e-liquid must not exceed 20mg/ml and e-liquid bottles must be limited in size to 10 ml;
- E-liquid bottles must be child-resistant and tamper evident;
- They must include new labelling requirements and warnings in local language;
- The e-liquid container must possess a securely attached nozzle at least 9 mm long;
- The e-liquid container must possess a flow control mechanism emits no more than 20 drops of refill liquid per minute;
- E-liquid advertising is prohibited;
- All products should be notified six months before the intended placing on the market.
Only nicotine-containing e-liquids must meet these requirements.

Child-proof and tamper-proof bottles

E-liquid bottles must be limited in size to 10 ml

The highest nicotine content allowed in an e-liquid is 20mg/ml

Bottles possess securely attached nozzle at least 9 mm long

Flow control mechanism emits no more than 20 drops of refill liquid per minute

A health warning, shall appear on 2 largest surfaces of the unit packet and any outside packaging and cover more than 30% of the surface of the unit packet
Requirements for ingredients
According to the TPD, e-liquids with nicotine cannot contain the following ingredients:
- Vitamins or other additives that create the impression that a tobacco product has a health benefit or presents reduced health risks.
- Caffeine or taurine or other additives and stimulant compounds that are associated with energy and vitality.
- Additives with colouring properties for emissions.
- Additives that have CMR (carcinogenic, mutagenic, reprotoxic) properties in unburnt form.
Our products do not contain banned ingredients.
Packaging and warnings
The new packaging of our products complies with the TPD requirements. All mandatory warnings and information appears on each unit packet.
Individual packaging must contain the following:
- Instructions for use and storage of the product;
- The amount of nicotine;
- The batch number;
- The following health warning that must be visible on two biggest planes of the packaging and cover more than 30% of the packaging area: “This product contains nicotine which is a highly addictive substance, it is not recommended to be sold to non-smokers.”
TPD notification
Manufacturers and importers of e-liquids must submit a notification to the competent authorities of any e-liquid which they intend to place on the market (for each flavor and nicotine level except for zero nicotine products).


Instructions for use and storage of the product

Nicotine content
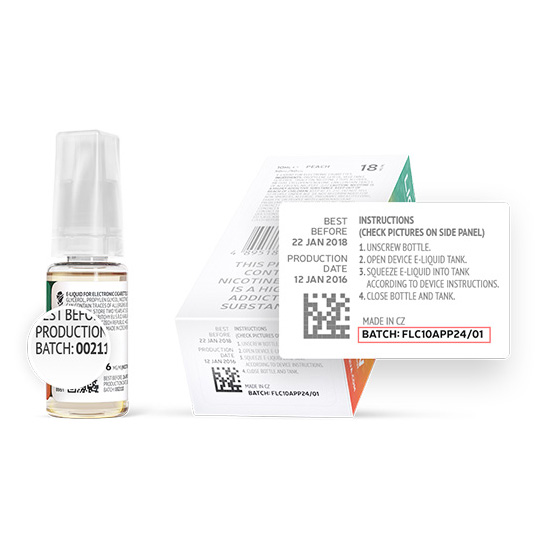
Batch number





